(b) PTPTB/PCBM + nile red in the dark (---) and under AM 1.5 conditions (——)
Linz Institute of Organic Solar Cells (LIOS), Physical Chemistry, University of Linz
Altenbergerstr. 69, 4040 Linz, Austria
*New address: Siemens AG, Central Research, CT MM1, D-91052, Erlangen
Stratingh Institute and Materials Science Center, University of Groningen
Nijenborgh 4, 9747 AG Groningen, The Netherlands
Laboratory of Macromolecular and Organic Chemistry, Eindhoven University of Technology
PO Box 513, 5600 MB Eindhoven, The Netherlands
Conjugated polymers attracted great interest as photovoltaic materials in the
recent years [1, 2]. Photoinduced electron transfer from conjugated polymers to fullerene
acceptors [3] and the bulk heterojunction concept [4] offer an efficient way for charge separation.
Consequent engineering of polymer/fullerene composite morphology and the contacts of the
devices resulted in an improvement of the power conversion efficiency of these solar cells up
to h > 2.5 % under AM1.5 illumination [5].
The limiting parameter of this system is the spectral mismatch between the absorption
(Eg > 2.2 eV) of the active layer and the terrestrial sunlight spectrum. For
efficient light harvesting, photovoltaic materials with bandgaps Eg < 1.8 eV are
needed. For improved spectral coverage of the available sunlight, additional absorbers for the
range < 2 eV have to be introduced.
In this contribution we present photovoltaic devices made of the novel low bandgap polymer
PTPTB [6,7], chemical structure is presented as insert in figure 1a, which has an absorption
maximum near the IR spectral region. Furthermore, we successfully sensitized this cell with
highly absorbing organic dye's. For the observed contribution of the organic dye to the
photocurrent, an energy transfer from the dye to the polymer is presumed.
Devices are prepared in sandwich configuration. ITO/PEDOT-PSS serves as anode
substrate and LiF/Al as top cathode. The active layers are spincast from polymer/PCBM 1/3
toluene solutions. The structure of PTPTB is shown as inset in figure 1a. For the dye sensitised
solar cells, 10-weight % dye is added to the polymer. The structure of Nile Red is shown as
insert in figure 1b.
 |
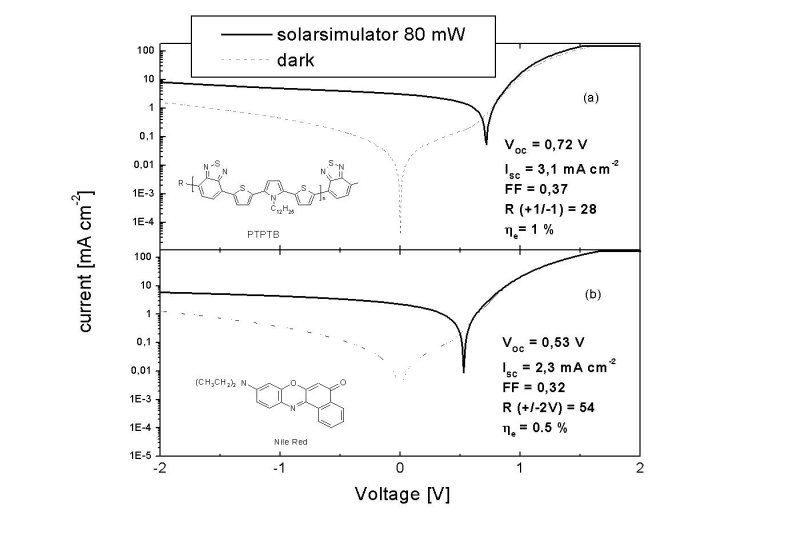
Figure 1 (a) I-V curve of a PTPTB/PCBM cell and (b) PTPTB/PCBM + nile red in the dark (---) and under AM 1.5 conditions (——) |
The bandgap of PTPTB is determined optically and electrochemically as ~1.6 eV. Figure 1a shows the I-V curve of a PTPTB/PCBM in the dark and under AM 1.5 simulated illumination. The open circuit voltage is just 100 mV below the one of MDMO-PPV cells, after a bandgap reduction of > 600 meV. The low fill factor may originate from the poor film quality giving rise to many shunts. Photoactivity of the polymer is shown with the IPCE measurements, figure 2a.
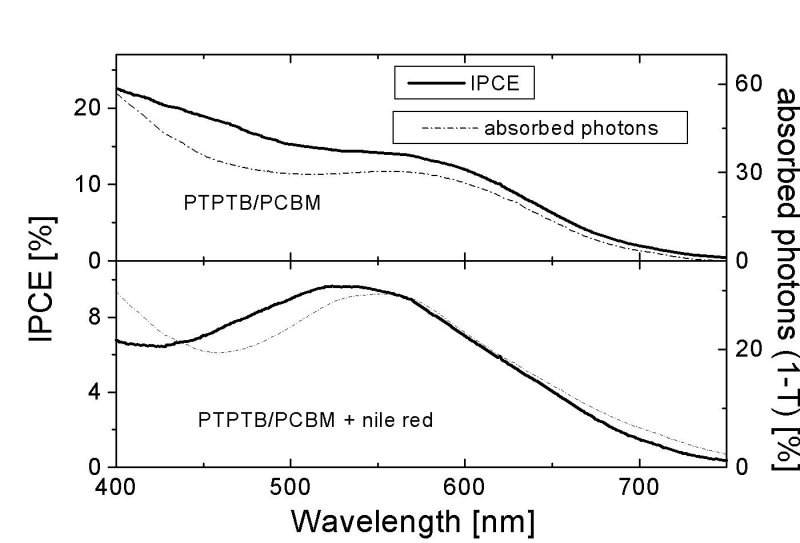 |
Figure 2 IPCE (——) and % of absorbed photons in transmission (---) of (a) PTPTB/PCBM and (b) PTPTB/PCBM + nile red |
Energy transfer in dye/polymer mixtures is investigated by photoluminescence
studies. In the two-component blend of the wide bandgap dye nile red, the structure is shown
as insert in figure 1b, and the low bandgap polymer PTPTB, the luminescence of the dye is
quenched, whereas the polymer luminescence is increased. After addition of PCBM, no
luminescence is detectable.
Devices from the three component blend shows high rectification and a clear photoeffect
under illumination, shown in figure 1b. The IPCE match the absorption spectrum.
References
| [1] | C. J. Brabec, N. S. Sariciftci, J. K. Hummelen, Advanced Functional Materials 11, No.1, 15 (2001). |
| [2] | M. Granström, K. Petritsch, A. C. Arias, A. Lux, M. R. Andersson, R. H. Friend, Nature 395, 257 (1998). |
| [3] | N. S. Sariciftci, L. Smilowitz, A. J. Heeger, F. Wudl, Science 258, 1474 (1992). |
| [4] | G. Yu, J. Gao, F. Wudl, A. J. Heeger, Science 270, 1789 (1995). |
| [5] | S. E. Shaheen, C. J. Brabec, N. S. Sariciftci, Applied Physics Letters 78, No. 6, 841 (2001). |
| [6] | A. Dhanabalan, P.A. Van-Hal, J.K.J. Van-Duren, J.L.J. Van-Dongen,R.A.J. Janssen, Synthetic Metals 119, No.1-3, 169 (2001). |
| [7] | A. Dhanabalan, P.A. Van-Hal, J.K.J. Van-Duren, J.L.J. Van-Dongen,R.A.J. Janssen, Advanced Functional Materials 11, No.4, p.255 (2001). |
email: christoph.winder@jku.at
hoem-page: www.ipc.uni-linz.ac.at